Case ID:
HJF 357-13
Web Published:
11/7/2025
Studies in humans and animals have demonstrated that infection with parasitic worms is protective against a range of chronic inflammatory diseases. These include inflammatory bowel disease, environmental allergies, asthma, and autoimmune diseases such as Type 1 diabetes, multiple sclerosis, and rheumatoid arthritis. Parasitic worm infections modulate host immune response by upregulating immune regulatory networks.
Scientists at the Uniformed Services University of the Health Sciences (USU) and the Henry M. Jackson Foundation (HJF) have discovered that the homogenate of Caenorhabditis elegans (C. elegans) worms grown in a clean, axenic media (free of living organisms other than C. elegans worms) can be used to create a safe and effective product to protect against hyperinflammatory diseases including autoimmunity and allergy.
Applications and Advantages
- Method to grow worms without the need for an animal host and to obtain large quantities of sterile material with adaptability to GMP manufacturing.
- Made without harmful bacterial toxins, ensuring safety and reducing risk of unwanted immune reactions.
- Flexibility in treating and managing an array of autoimmune diseases including type 1 diabetes, multiple sclerosis, rheumatoid arthritis, Crohn’s disease, and others linked to immune system imbalance.
- Allows development of multiple dose formulations and routes of administration for long-term use with repeated treatments.
- Derived from a natural source with potential for use as a dietary supplement or functional food ingredient.
- Proven in animal studies to delay disease onset and reduce inflammation, showing real potential benefits.
Innovation Description
While parasitic worm infections have been used successfully to treat inflammatory bowel disease, there are a number of factors limiting their translation into widespread clinical use. These include difficulty in cultivating parasitic worms in sterile conditions in a laboratory environment and associated high expenses, difficulty in obtaining sufficient material for patient treatment, potential difficulties ensuring homogeneity of parasitic worms from batch-to-batch, risk of pathology induced from live worm infections, and potential unease of patients with regards to being infected with parasitic worms.
The novel technology developed by HJF and USU researchers solves most of the problems associated with the use of parasitic worms as a therapeutic. This method enables cultivation of large quantities of nematodes (round worms) under sterile, laboratory-controlled conditions. Specifically, the technology involves growing C. elegans worms in a special lab-made axenic media free of any bacteria to generate the axenic C. elegans (aCeAg) homogenate. When administered to animals prone to autoimmune disease, the aCeAg helped increase protective immune signals and lowered harmful inflammation.
These studies demonstrate that twice weekly intraperitoneal injections of aCeAg confer substantial protection against type 1 diabetes mellitus in the NOD mouse model. In contrast to the 80% of control (PBS-injected) NOD mice that developed type 1 diabetes, only 10% of aCeAg-treated NOD mice developed clinical disease (Fig 1). Further, at study endpoint, aCeAg-treated mice exhibited significantly greater numbers of insulin-producing pancreatic islets and greater numbers of uninfiltrated islets. These studies demonstrate that aCeAg is as potent as live worms in treating autoimmunity in animal models.
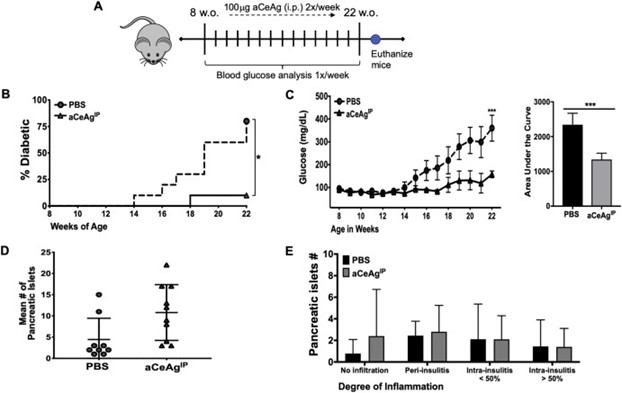
Fig. 1 aCeAg delays the onset of Type 1 Diabetes pathology in NOD mice.
Furthermore, this technology is well-suited for potential development as a food additive or stand-alone oral treatment for multiple chronic diseases targeted by the nutraceutical, such as irritable bowel syndrome, celiac disease, and allergy. In addition, this technology can be developed for veterinary use for multiple, difficult to treat diseases in animals, such as dermatitis in canines. As worms are considered a "natural product” there may be fewer hurdles involved in development of this technology into a variety of nutraceutical-like products for humans and animals.
Inventors
- Edward E. Mitre, M.D. (USUHS)
- Marina Torrero, Ph.D. (HJF)
- Belinda Jackson Ph.D. (HJF)
Innovation Status
In vivo data is available.
Please see J Transl Autoimmun. 2020 Aug 16;3:100065. doi: 10.1016/j.jtauto.2020.100065. PMID: 32939449; PMCID: PMC7476867 .
Intellectual Property Status
Issued patents in CA, AU, US, FR, DE, GB.
